Abstract
Background: Multiple myeloma (MM) is a malignant plasma cell disorder that is characterized by osteolytic lesions, increased susceptibility to infections, hypercalcemia, anemia, and renal failure. For patients with newly diagnosed MM (NDMM), autologous stem cell transplant (ASCT) is an important treatment strategy and has demonstrated significant improvement in progression-free survival (PFS). Maintenance treatment after ASCT has offered prolonged disease control and improved outcomes, and lenalidomide (Len) is now considered the standard of care as recommended by NCCN and ESMO guidelines. However, median PFS on Len maintenance (mean duration of treatment: approximately 2-3 years) is only 52.8 months, long-term tolerability is poor, and most patients eventually relapse. Therefore, there is a need for new therapeutic options in the maintenance setting that may provide improved PFS and overall survival (OS). Teclistamab (Tec) is a B-cell maturation antigen (BCMA) × CD3 bispecific antibody that redirects CD3+ T cells to induce cytotoxicity towards BCMA-expressing MM cells. In the phase 1/2 MajesTEC-1 study (NCT03145181), Tec monotherapy was well tolerated and showed encouraging efficacy, yielding durable responses that deepened over time in heavily pretreated patients with relapsed/refractory MM. The combination of Tec + Len (Tec-Len) is being investigated in the MajesTEC-2 study in patients with MM who have received ≥2 prior lines of therapy (LOT), including exposure to a proteasome inhibitor (PI), an immunomodulatory drug (IMiD), and an anti-CD38 monoclonal antibody (NCT04722146). Here we report the design of the randomized, open-label, multicenter, phase 3 MajesTEC-4 trial (NCT05243797), which will compare the efficacy of Tec-Len versus Len alone as maintenance therapy in patients with NDMM who have completed induction treatment followed by ASCT, with or without consolidation. As studies have suggested that IMiDs may enhance or improve the efficacy of immunotherapies, it is anticipated that the combination of Tec-Len in the maintenance setting will provide greater efficacy than Len alone.
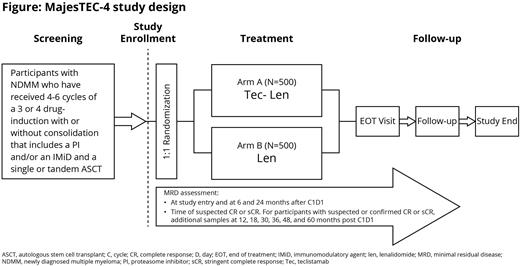
Study Design and Methods: Eligible patients will be aged ≥18 years with NDMM (per International Myeloma Working Group [IMWG] criteria) who received 4-6 cycles of 3 or 4 drug-induction treatments (only 1 LOT, including a PI and/or IMiD with or without an anti-CD38 monoclonal antibody and achieved ≥partial response [PR]), had a single or tandem ASCT, and had an Eastern Cooperative Oncology Group performance status score 0-2. Patients who received any prior BCMA-directed treatment or have progressive disease will be excluded. As the combination of Tec-Len has not been previously evaluated in patients with NDMM, a safety run-in will be performed. If no safety signal is observed, randomization will begin. Approximately 1000 patients will be randomized 1:1 to receive 28-day cycles of Tec-Len or Len. The study will continue until ~380 deaths have occurred or a maximum of 8 years after the last patient is randomized. The primary endpoint is PFS. Secondary endpoints include complete response (CR) conversion from PR/very good PR, minimal residual disease (MRD) negative conversion, ≥CR, MRD negative CR, sustained MRD negativity, OS, PFS2, time to next treatment, patient-reported outcomes, and incidence and severity of adverse events (AEs). Response will be assessed using 2016 IMWG criteria. AEs will be graded by Common Terminology Criteria for AEs v5.0, except for cytokine release syndrome and immune effector cell-associated neurotoxicity syndrome, which will be graded by American Society for Transplantation and Cellular Therapy guidelines. The study opened for enrollment in May 2022. Results from this trial will provide insights into a possible new synergistic combination that could improve response and prolong survival versus current maintenance regimens for patients with NDMM.
Disclosures
Zamagni:janssen, bms, amgen, roche, pfizer, sanofi, gsk: Honoraria, Other. Boccadoro:Bristol Myers Squibb: Honoraria, Research Funding; Novartis: Honoraria, Research Funding; Janssen: Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Amgen: Honoraria, Research Funding; Celgene: Honoraria, Research Funding; Sanofi: Honoraria, Research Funding; AbbVie: Honoraria; GlaxoSmithKline: Membership on an entity's Board of Directors or advisory committees; Mundipharma: Research Funding. Spencer:Amgen: Consultancy, Honoraria; Haemalogix: Consultancy, Honoraria; Janssen: Consultancy, Honoraria, Research Funding, Speakers Bureau; BMS: Consultancy, Honoraria; AbbVie: Consultancy, Honoraria, Research Funding, Speakers Bureau; Roche: Honoraria, Membership on an entity's Board of Directors or advisory committees; Pfizer: Consultancy, Honoraria. Delforge:Janssen: Honoraria, Speakers Bureau; BMS: Honoraria; Amgen: Honoraria; GSK: Honoraria. Reece:Karyopharm: Consultancy; BMS: Research Funding; Millenium: Research Funding; Amgen: Consultancy, Honoraria; Celgene: Consultancy, Honoraria, Research Funding; Merck: Research Funding; Takeda: Consultancy, Honoraria, Research Funding; Janssen: Consultancy, Honoraria, Research Funding; Otsuka: Research Funding; Sanofi: Honoraria; GSK: Honoraria. Gyula Szabo:Takeda: Consultancy; Janssen: Consultancy. Einsele:BMS/Celgene: Consultancy, Membership on an entity's Board of Directors or advisory committees, Other: Travel Grants, Research Funding; Janssen: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Other: Travel Grants, Research Funding; Amgen: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Other: travel grants; Takeda: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Other: travel grants; Sanofi: Consultancy, Honoraria, Research Funding; GSK: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; Novartis: Other: travel grants. Terpos:EUSA Pharma: Honoraria, Other: Travel expenses; Novartis: Honoraria; Janssen: Honoraria, Research Funding; Genesis: Honoraria, Research Funding; Amgen: Honoraria, Other: Travel expenses, Research Funding; GSK: Honoraria, Research Funding; BMS: Honoraria; Takeda: Honoraria, Other: Travel expenses, Research Funding; Sanofi: Honoraria, Research Funding. Schjesvold:AbbVie: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Novartis: Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Targovax: Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Celgene: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Janssen-Cilag: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Bristol Myers Squibb: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Amgen: Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Sanofi: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Skylite DX: Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Takeda: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Daiichi Sankyo: Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Oncopeptides: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; GlaxoSmithKline: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Pfizer: Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding. Bila:Janssen: Consultancy, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; AMICUS representing AMGEN: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Takeda: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau. Beksac:Sanofi: Consultancy, Honoraria, Other: Advisory Boards, Speakers Bureau; Amgen: Consultancy, Honoraria, Other: Advisory Boards, Speakers Bureau; Takeda: Consultancy, Honoraria, Other: Advisory Boards, Speakers Bureau; Janssen: Consultancy, Honoraria, Other: Advisory Boards, Speakers Bureau; Oncopeptides: Consultancy, Honoraria, Other: Advisory Boards, Speakers Bureau. Cook:Takeda, BMS, Amgen, Roche, Janssen, Sanofi, Karyopharm, Pfizer: Consultancy; Takeda, BMS: Research Funding; Takeda, BMS, Amgen, Janssen, Sanofi: Speakers Bureau. Rodriguez:Janssen, BMS, Takeda, AbbVie, karyopharm, Artiva: Consultancy, Speakers Bureau. Pei:Janssen R&D: Current Employment, Current equity holder in private company, Current holder of stock options in a privately-held company. Shi:Janssen: Current Employment. Sakabedoyan:Janssen- Johnson & Johnson: Current Employment, Current equity holder in private company. Jasielec:Janssen: Current Employment, Current equity holder in publicly-traded company. Amin:Janssen Research & DeveloJanssen Research & Development, LLCpment, LLC: Current Employment, Current equity holder in private company, Current holder of stock options in a privately-held company. Kobos:Janssen: Current equity holder in private company, Current holder of stock options in a privately-held company. Sonneveld:Janssen: Membership on an entity's Board of Directors or advisory committees, Research Funding; Amgen: Membership on an entity's Board of Directors or advisory committees, Research Funding; Celgene: Membership on an entity's Board of Directors or advisory committees, Research Funding; Pfizer: Membership on an entity's Board of Directors or advisory committees; Bristol Myers Squibb: Membership on an entity's Board of Directors or advisory committees, Research Funding; Karyopharm: Membership on an entity's Board of Directors or advisory committees, Research Funding. Van De Donk:Cellectis: Research Funding; Servier: Membership on an entity's Board of Directors or advisory committees; Amgen: Membership on an entity's Board of Directors or advisory committees, Research Funding; Adaptive Biotechnologies: Membership on an entity's Board of Directors or advisory committees; Celgene: Membership on an entity's Board of Directors or advisory committees, Research Funding; Bristol Myers Squibb: Membership on an entity's Board of Directors or advisory committees, Research Funding; Novartis: Membership on an entity's Board of Directors or advisory committees, Research Funding; Roche: Membership on an entity's Board of Directors or advisory committees; Bayer: Membership on an entity's Board of Directors or advisory committees; Takeda: Membership on an entity's Board of Directors or advisory committees; Janssen Pharmaceuticals: Membership on an entity's Board of Directors or advisory committees, Research Funding.
OffLabel Disclosure:
at the time of abstract submission, teclistamab is being investigated for the treatment of multiple myeloma but is not yet not approved
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal